The US Centers for Disease Control and Prevention issued an advisory Tuesday about the risks of counterfeit or mishandled Botox injections after dangerous fake versions of the product have been found in several states.
The CDC, the US Food and Drug Administration and state and local officials are working to investigate clusters of at least 22 women who have reported harmful reactions after receiving counterfeit Botox injections, injections from unlicensed or untrained providers or shots in places that were not health-care settings, such as spas or private homes, according to the official CDC health advisory.
As of Thursday, those incidents have been reported in California, Colorado, Florida, Illinois, Kentucky, Nebraska, New Jersey, New York City, Tennessee, Texas and Washington. Among the people who reported symptoms, 11 were hospitalized, the CDC said, but none have died.
The patients, ages 25 to 59, reported their symptoms between November and March, and most of them – 91% – reported receiving Botox for cosmetic purposes, according to the CDC. Their symptoms included blurred vision, drooping eyelids, dry mouth, slurred speech, shortness of breath, fatigue and weakness. Seven of them were tested for botulism. Test results were negative for six, while one is still pending.
These incidents appear to be connected to products that have been purchased from unlicensed sources and then administered by either unlicensed or licensed providers, according to a previous FDA announcement. The agency warned health-care professionals that purchasing and administering counterfeit products puts patients’ health at risk.
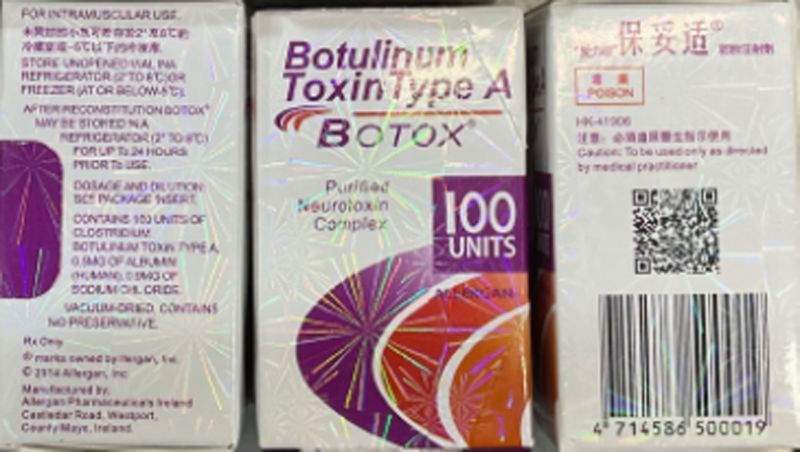
The counterfeit Botox products may be identified by the lot number C3709C3 on the outer carton and vial, according to the FDA.
The outer carton also may display the active ingredient as “Botulinum Toxin Type A” instead of “OnabotulinumtoxinA.” The carton and vial may indicate 150-unit doses, which is not a dosage made by the companies AbbVie or Allergan, the FDA said, and the outer carton may contain language that is not English.
In Tuesday’s advisory, the CDC noted that when botulinum toxin, known as Botox, diffuses around the site where it was injected, it can result in adverse effects.
“Botulism is the disease caused by botulinum toxin circulating in the blood and producing effects remotely from the injection site. There may be symptom overlap between the presentation of localized adverse effects from injection of botulinum toxin, especially in the head and neck, and the early symptoms of botulism,” according to the advisory.
“Clinicians and health departments should consider the possibility of adverse effects from botulinum toxin injections in patients presenting with localized paralysis,” the advisory says. “Clinicians should immediately contact their state, tribal, local, or territorial health department if they suspect botulism.”
Botulism can be treated with an antitoxin, according to the CDC. That’s why it’s important to get medical care quickly and early in the course of the disease. If the disease is left untreated, it may progress to paralysis and other complications.
“Botulinum toxin should be administered only by licensed providers, using only recommended doses of FDA-approved botulinum toxin, preferably in a licensed or accredited healthcare setting,” the CDC advisory says. “Providers should be trained in the proper administration of botulinum toxin, practicing in accordance with state and local requirements.”
In response to the reports of counterfeit Botox, the companies behind the FDA-approved versions, AbbVie and Allergan Aesthetics, said in a statement that they are the only authorized suppliers of Botox in the United States.
“Allergan Aesthetics, an AbbVie company, has a comprehensive supply chain security program to ensure that all products manufactured are safe, secure, and sold through authorized distribution channels. All reports of counterfeit product are thoroughly investigated by our team and in conjunction with law enforcement and public health authorities where appropriate,” the statement says.
Providers and consumers can identify authentic Botox products by a tamper-evident seal on the packaging, the name “onabotulinumtoxinA” listed under the brand name Botox and a hologram of the word “Allergan” on the vial label.
Botox is a brand name for botulinum toxin, which in small doses can temporarily treat medical or cosmetic concerns such as severe underarm sweating, chronic migraines, overactive bladder and facial wrinkles.
“Botox and its peer brands provide a dilute, precise, and consistent amount of botulinum toxin, whereas knock-off unregulated products are not reliably manufactured or subject to the same quality assurance, which can lead to over or underdosing,” Dr. Michael Cameron, president and founder of Cameron Dermatology and assistant clinical professor of dermatology at Mount Sinai Health System in New York, said last week.
When in the body at high levels, the toxin botulinum can cause the rare but serious illness botulism or symptoms similar to those from the illness.
“Signs and symptoms of botulism include muscle weakness, vision changes, slurred speech, difficulty moving your eyes. An emergency would be if you start to have difficulty breathing,” Cameron said.
“I personally recommend if you’re getting Botox for your wrinkles, you go to a board-certified dermatologist. If you’re getting Botox for your migraines, you go to a neurologist. If you’re getting Botox for your bladder issues, you go to your urologist,” he said. “If you feel like there’s any risk or the pricing doesn’t really make sense, then you could just ask to actually see the vial and inspect the vial of Botox yourself.”